The New Features of the Latest iGrafx 17.4 Release

While some companies talk about their “open door” and “transparency” with their customers, at iGrafx, those are part of our core values. A large percentage of updates and enhancements you have seen to our platform over the years have come directly from you. Our latest 17.4 release is no exception.
In addition to updated and enhanced web modelling capabilities, the latest release of iGrafx 17.4 provides greater capabilities for managing platform objects and increased audit capabilities requested by many of our customers.
Who can create?
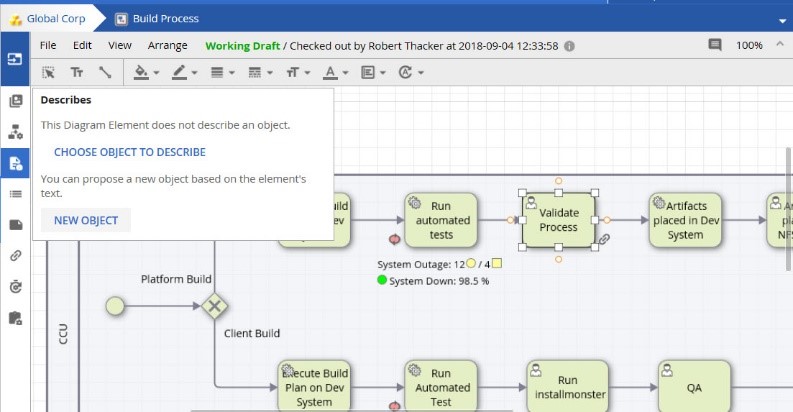
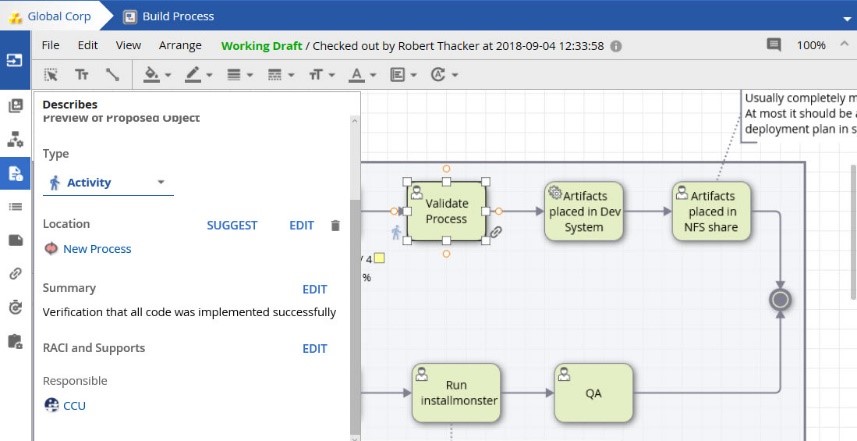
In the past, the creation of platform objects was restricted to only those with Architect access. In 17.4 this capability has been extended, so that those who are responsible for diagramming can now propose new objects into the process landscape.
These Designers can also propose the location and RACI (Responsible, Accountable, Consulted, Informed) relationship information, thus expediting the creation of our modeling and process landscape.
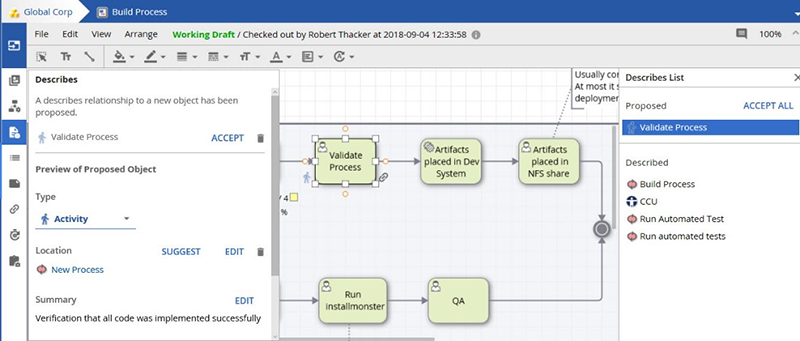
Once created, Architects can review, correct, and accept any new proposals.
Improved Audit Capabilities
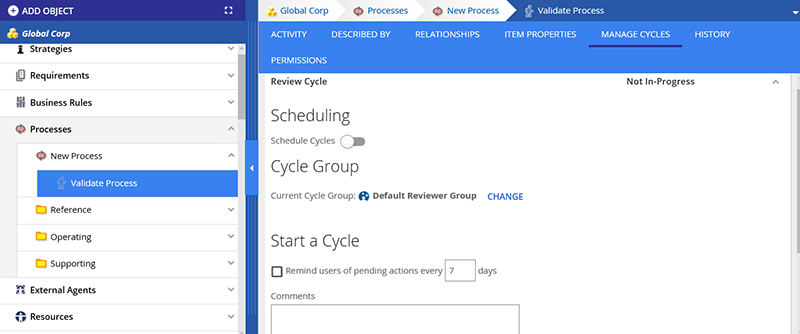
It has been requested on numerous occasions that the cycle management capabilities of documents and diagrams be extended to all objects within the platform. In the latest 17.4 release, this is now possible. All objects can now be submitted for OR have a defined scheduled review cycle.
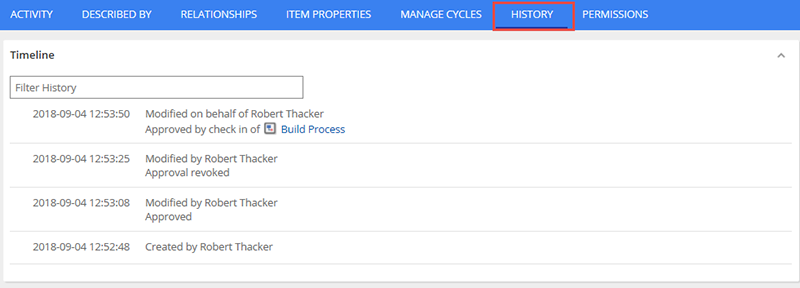
All updates to the object are also tracked so that auditors may see when items were last modified, reviewed, approved, and by who.
In addition, you will now be able to submit multiple objects for review or approval at the same time. This will greatly increase the efficiency of those that need to schedule multiple files or even entire directories for approval at the same time.
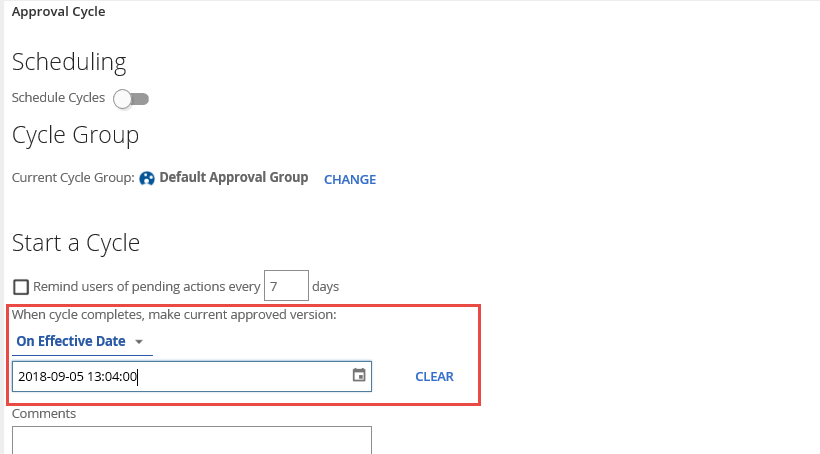
In some instances, while the document may be approved, the actual effective date of the new policy or process may be in the future.
Having this ability provides the option of revising and getting the documents approved prior to the updates going live. This effective date on documentation is also a requirement in some Health Care, Pharmaceutical, and Manufacturing audits.
Your cycle management settings can be reported on so that you can quickly view not just current, but upcoming cycles.
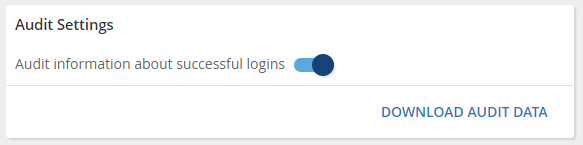
This will provide greater visibility into who is accessing the platform and how it is being used.
Would you like to see these updates in action? Check out this video highlighting the changes and get a complete list 17.4 updates and enhancements in our release notes.